|
Thus, gaseous hydrogen is not absorbed by metals at ambient temperatures, as it is in molecular form, in which pairs of atoms are tightly bound together. 
Hydrogen is normally only able to enter metals in the form of atoms or hydrogen ions. 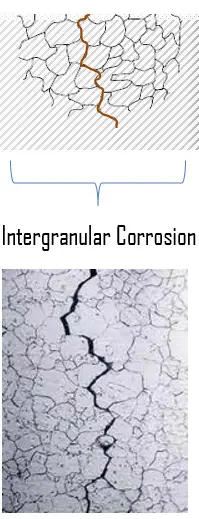
Click here to see our latest technical engineering podcasts on YouTube. The severity of hydrogen embrittlement is a function of temperature: most metals are relatively immune to hydrogen embrittlement, above approximately 150☌. Such stress states can be caused both by the presence of residual stresses, associated fabrication operations such as forming and welding, and applied service stresses. This happens when sufficient stress is applied to a hydrogen-embrittled object. The phenomenon usually becomes significant when it leads to cracking. Microstructures which bestow high strength, often monitored by hardness level, or having specific distributions of grain boundary particles or inclusions, can result in increased susceptibility to embrittlement. The degree of embrittlement is influenced both by the amount of hydrogen absorbed and the microstructure of the material. 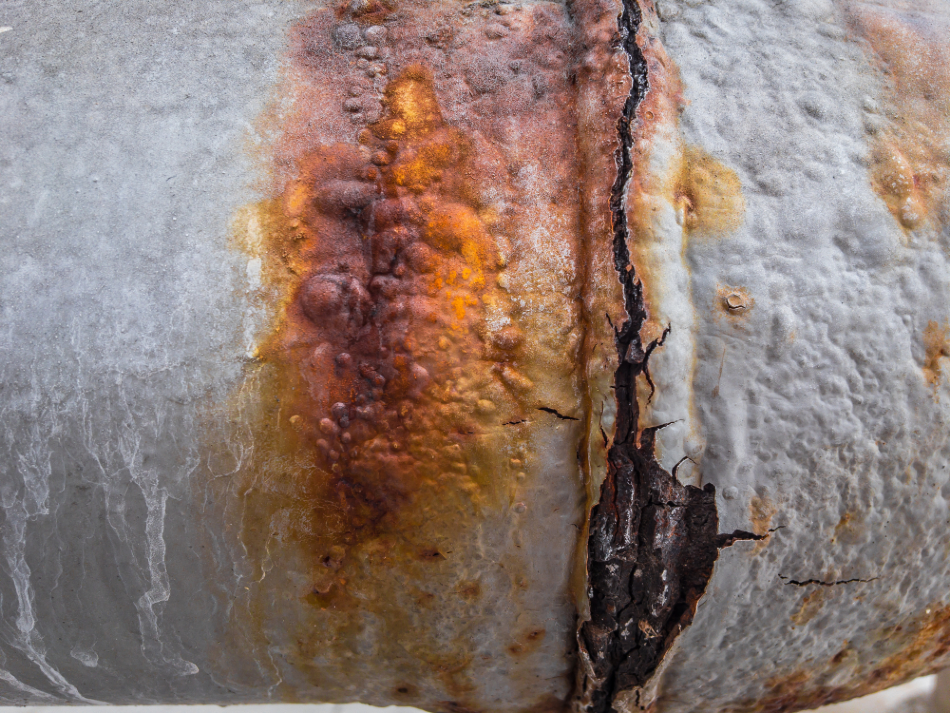
Hydrogen Embrittlement occurs when metals become brittle as a result of the introduction and diffusion of hydrogen into the material.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
